Cortisporin-TC Otic
Cortisporin-TC Otic (colistin sulfate – neomycin sulfate – thonzonium bromide – hydrocortisone acetate otic suspension)
Product Summary
Therapeutic Class: Ear Nose & Throat Preparations
This product is manufactured and distributed by MCP Pharmaceuticals, LLC
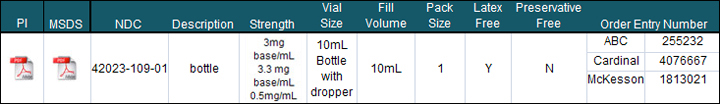
Product Profile:
- Latex Free
- Bar Coded
- Otic Preparation
Description
Cortisporin -TC Otic Suspension is a sterile anti-bacterial and anti-inflammatory aqueous suspension containing in each mL:
- Colistin base activity, 3mg (as the sulfate)
- Neomycin base activity, 3.3 mg (as the sulfate)
- Hydrocortisone acetate, 10 mg (1%)
- Thonzonium bromide, 0.5 mg (0.05%)
- Polysorbate 80, acetic acid and sodium acetate in a buffered aqueous vehicle
- Thimerosol (mercury derivative) 0.002% is added as a preservative
It is a non-viscous liquid, buffered at pH 5, for instillation into the canal of the external ear or direct application to the affected aural skin.
Indications and Usage
For the treatment of superficial bacterial infections of the external auditory canal, caused by organisms susceptible to the action of the antibiotics; and for the treatment of infections of mastoidectomy and fenestration cavities, caused by organisms susceptible to the antibiotics.
Contraindications
This product is contraindicated in those individuals who have shown hypersensitivity to any of its components. This product should not be used if the external auditory canal disorder is suspected or known to be due to cutaneous viral infection (e.g., herpes simplex virus or varicella zoster virus).
Warnings
Neomycin can induce permanent sensorineural hearing loss due to cochlear damage, mainly destruction of hair cells in the organ of Corti. The risk is greater with prolonged use. Therapy should be limited to 10 consecutive days. (See PRECAUTIONS-General section of Prescribing Information). Patients being treated with eardrops containing neomycin should be under close clinical observation. Cortisporin® -TC Otic Suspension should be used cautiously in any patient with a perforated tympanic membrane. Neomycin sulfate may cause cutaneous sensitization. A precise incidence of hypersensitivity reactions (primarily skin rash) due to topical neomycin is not known. Discontinue promptly if sensitivity or irritation occurs.
When using neomycin-containing products to control secondary infection in the chronic dermatoses, such as chronic otitis externa or stasis dermatitis, it should be borne in mind that the skin in these conditions is more liable than is normal skin to become sensitized to many substances, including neomycin. The manifestation of sensitization to neomycin is usually a low-grade reddening with swelling, dry scaling, and itching; it may be manifest simply as a failure to heal. Periodic examination for such signs is advisable, and the patient should be told to discontinue the product if they are observed. These symptoms regress quickly on withdrawing the medication. Neomycin-containing applications should be avoided for the patient thereafter.
Adverse Events
Neomycin occasionally causes skin sensitization. Ototoxicity (see WARNINGS section of the Prescribing Information) and nephrotoxicity have also been reported.
Adverse reactions have occurred with topical use of antibiotic combinations.
Exact incidence figures are not available since no denominator of treated patients is available. The reaction occurring most often is allergic sensitization. In one clinical study, using a 20% neomycin patch, neomycin-induced allergic skin reactions occurred in two of 2,175 (0.09%) individuals in the general population. In another study the incidence was found to be approximately 1%.
The following local adverse events have been reported with topical corticosteroids especially under occlusive dressings: burning, itching, irritation, dryness, folliculitis, hypertrichosis, acneiform eruptions, hypopigmentation, perioral dermatitis, allergic contact dermatitis, maceration of the skin, secondary infection, skin atrophy, striae, and miliaria.